Generation of human CD4+ T-regs:
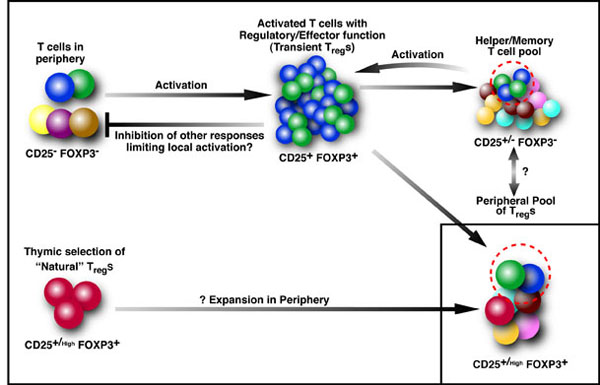
In addition to dissecting CD8+ T cell-mediated immune regulation, we are also interested in the immune biology of CD4+CD25+FOXP3+ regulatory T cells (T-regs). These cells form an important arm of the immune system responsible for suppressing untoward immune responses. T-regs can be thymically derived or peripherally induced, even from CD4+CD25-FOXP3- T-cells. FOXP3 expression and in vitro suppressive activity are considered unique hallmarks of this dedicated and stable lineage of regulatory cells. However, our recent studies show that virtually all human CD4+CD25-FOXP3- T-cells and CD8+CD25-FOXP3- T-cells attain a transient FOXP3+CD25+ state during activation. In this state of activation, these cells possess the classic phenotype of T-regs, in that they express similar markers and inhibit in vitro proliferation of autologous CD4+CD25- T-cells. This state is characterized by suppressed IFN-γ production and robust TNF-α and IL-10 production. Interestingly, the great majority of the activated cells eventually downregulate FOXP3 expression, with a concomitant drop in suppressive ability. Thus, in humans, FOXP3 expression and T-reg functionality are not exclusive features of a stable or unique lineage of T-cells, but may also be a transient state attained by almost all T-cells. These results warrant caution in interpreting human studies using FOXP3 and suppressive activity as readouts and suggest that attempts to induce “T-regs” may paradoxically result in induction of effector T-cells, unless stability is confirmed. Such induced T-regs and their role in various disease situations are a focus of several projects in the lab.
CD4 effector resistance to CD4 and CD8-mediated regulation:
T-cell-driven immunopathology is a central feature of multiple immune-mediated diseases. As a consequence, enhancing the immune regulation of pathogenic T-cell responses is an important quest in the approach to autoimmune diseases. Our group has had a long-standing interest in understanding the immune process that underlie the pathogenesis and regulation of autoimmunity, with a focus on multiple sclerosis (MS). Through studies in MS and its animal model (EAE), we have demonstrated the critical disease regulatory role of CD8 T-cells in disease. Similar appreciation for the role of these cells in downregulating other autoimmune diseases has also emerged. At the same time, it is also becoming clear that severe autoimmune disease is also characterized by increased resistance of effector/pathogenic CD4 T-cells to immune regulation. Our studies demonstrate several novel aspects of the CD4-CD8 effector-regulator dynamics and based on these preliminary findings, we hypothesize that maturation of CD4 T-helper cells along different lineages results in differential levels of effector resistance. Similarly, maturation of CD8 T-cells under differing conditions results in differential regulatory ability. We further hypothesize that these resistant/regulatory phenotypes are plastic in nature, in that they can be modulated in memory and effector cell populations by appropriate stimuli. We are addressing these central hypotheses in ongoing experiments focused on: (1) Determining the role of CD4 T-helper cell differentiation and effector resistance, with a particular focus on the mechanisms of resistance of Th17 cells and the plasticity of memory CD4 T-cells; and (2) Understanding the modulation of CD8 T-cell suppressor ability, with a focus on different CD8 lineages as well as CD4-CD8 cross-talk.
These studies address a poorly understood but fundamental feature of the interaction between effector CD4 T-cell responses and their immune regulators, in the context of the human immune system. The results will have clear implications not only for the therapeutic approach to autoimmune diseases, but also in the understanding of multiple clinical situations where immune interactions play a critical role in determining outcome (infectious diseases, allergy, cancer, to name a few).
Learn More:
Transient regulatory T-cells: a state attained by all activated human T-cells. Pillai V, Ortega SB, Wang CK, Karandikar NJ. Clin Immunol. 2007 Apr;123(1):18-29. Epub 2006 Dec 19. PMID:17185041
Human regulatory T cells: a unique, stable thymic subset or a reversible peripheral state of differentiation? Pillai V, Karandikar NJ. Immunol Lett. 2007 Nov 30;114(1):9-15. Epub 2007 Sep 29. Review. PMID:17945352
Attack on the clones? Human FOXP3 detection by PCH101, 236A/E7, 206D, and 259D reveals 259D as the outlier with lower sensitivity. Pillai V, Karandikar NJ. Blood. 2008 Jan 1;111(1):463-4; author reply 464-6. No abstract available. PMID: 18156502